The African Union (AU) is making good progress in operationalising the African Medicines Agency (AMA), having recently formed its Bureau and currently shaping its governing board.
This is according to Chimwemwe Chamdimba, Head of Health at the AU Development Agency (AUDA-NEPAD), who addressed a session of the Africa Health Agenda International Conference (AHAIC) in Rwanda on Wednesday.
AMA is being set up as a specialised health agency of the AU to ensure the regulatory harmonisation of medicines across Africa.
“We are hopeful that, in the next quarter, we should have the governing board of the AMA formalised by the congress of state parties,” Chamdimba told the session on the regulatory harmonisation, which was organised by the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA).
Tackling fragmentation
“As the African Union, from the very beginning our thoughts have been to move from the fragmented 55 member states’ regulatory authorities, where manufacturers have to submit their [product] dossiers to each regulator, to the five regional economic communities (RECs),” said Chamdimba.

She says that the RECs started harmonisation efforts as early as 2009, and the aim is to develop a common product application template that can be used by all the countries in the region.
By working together in the regions, national regulators had been able to do joint assessments and inspections, and this has started to “create trust between the regulator authorities within the RECs”, she added.
The AU is currently developing a continental regulatory reliance framework that will be piloted in the East Africa region.
In addition, the AU was working with partners such as Amref to train national regulators.
AUDA Nepad has also been mandated by the AU to lead private sector engagement in health and “we want as much as we can to have a more systematic way of engaging the private sector”, she added.
“We all need to hold hands. The proof of the pudding is in the eating and we can only get feedback from the industry if they are willing to try the processes that we are putting in place on the continent.”
Timely access to medicines
Johnson & Johnson’s Jacqueline Acquah, the IFPMA’s co-chair of the Africa regulatory network, said that AMA was essential to ensure that patients had timely access to medicines.
“As a result of the globalisation of markets, we have a lot of products that move in within international commerce, and supply chains have become very, very complex,” said Acquah.
“The public has become more aware, and there’s a high expectation of our health systems. Unfortunately, the world over there’s a limited resource when it comes to global regulatory affairs. Regulatory resources are very, very limited so it’s important that national regulatory agencies collaborate more.”
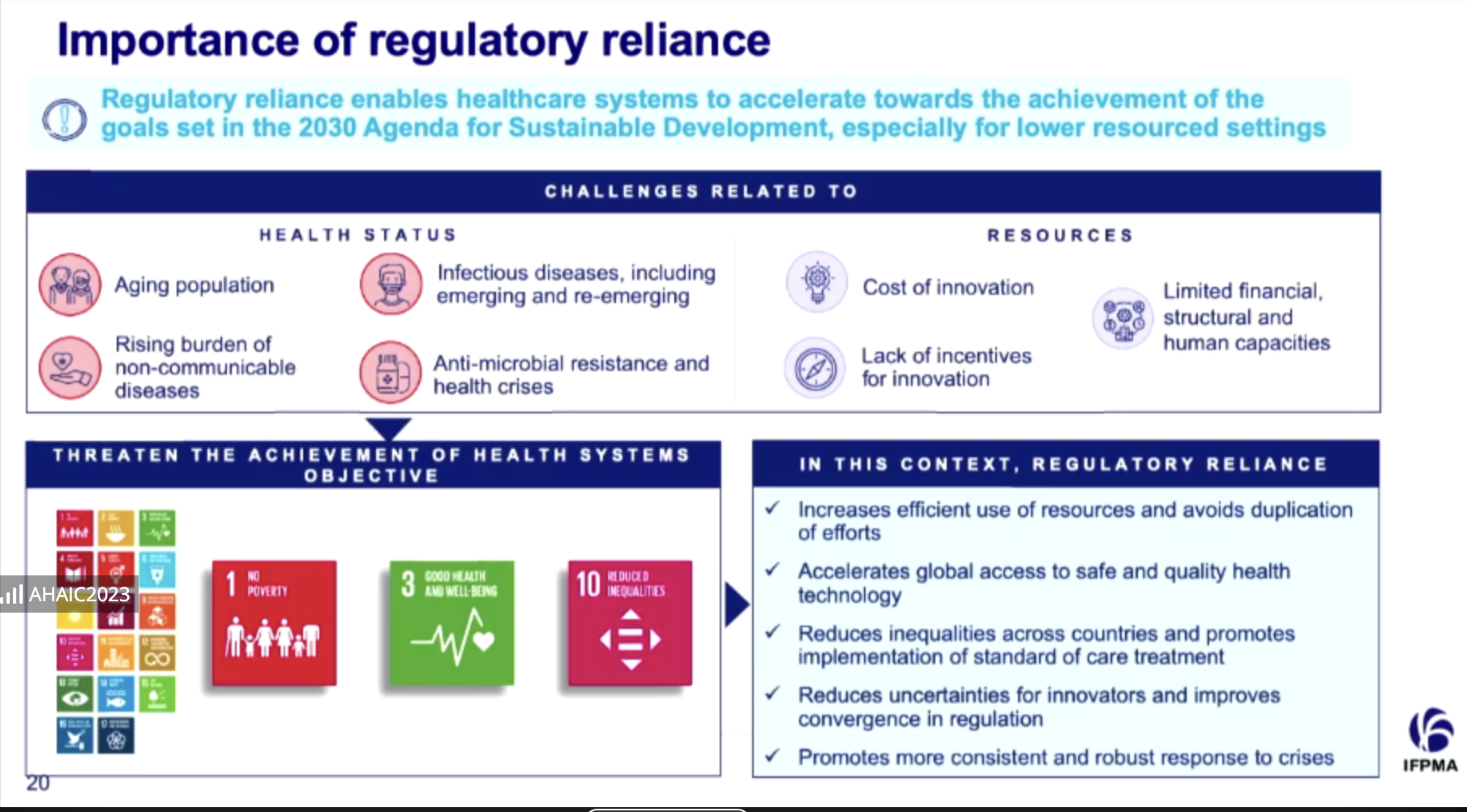
As a result, said Acquah, regulators needed to develop a reliance on one another to avoid duplication of work and resources.
“Regulatory reliance is when a regulator in one jurisdiction gives significant weight to the work that has been done by another regulator in another jurisdiction or an agency such as the World Health Organization (WHO) when making its own decisions,” she said.
“The regulatory agency is, however, still sovereign in the decision-making process, and also accountable for the decisions that they make.”
Egypt Drug Administration’s Dr Asmaa Fouad described regulatory reliance as “one of the magic tools that help regulators and industry and patient at the same time, so we achieve a triple win situation”.









